|
As news of his remarkable accomplishment began to spread, Mendeleev became something of a hero, and interest in the periodic table soared. The Periodic Table of Element an arrangement of 118 chemical element by Mendeleev that has spun our kids through generations. In a prestigious Faraday Lecture to the Royal Institution in London in 1889, he admitted that he had not expected to live long enough ‘to mention their discovery to the Chemical Society of Great Britain as a confirmation of the exactitude and generality of the periodic law’. Scandium and Germanium were the other two elements discovered by 1886, and helped to cement the reputation of Mendeleev’s periodic table. Mendeleev himself was surprised by how fast his ideas were confirmed. How did Mendeleev contribute to the periodic table The table below shows the example of Gallium, which Mendeleev called eka-aluminium, because it was the element after aluminium. Within twenty years, all three had been found, and their properties confirmed his predictions almost exactly. 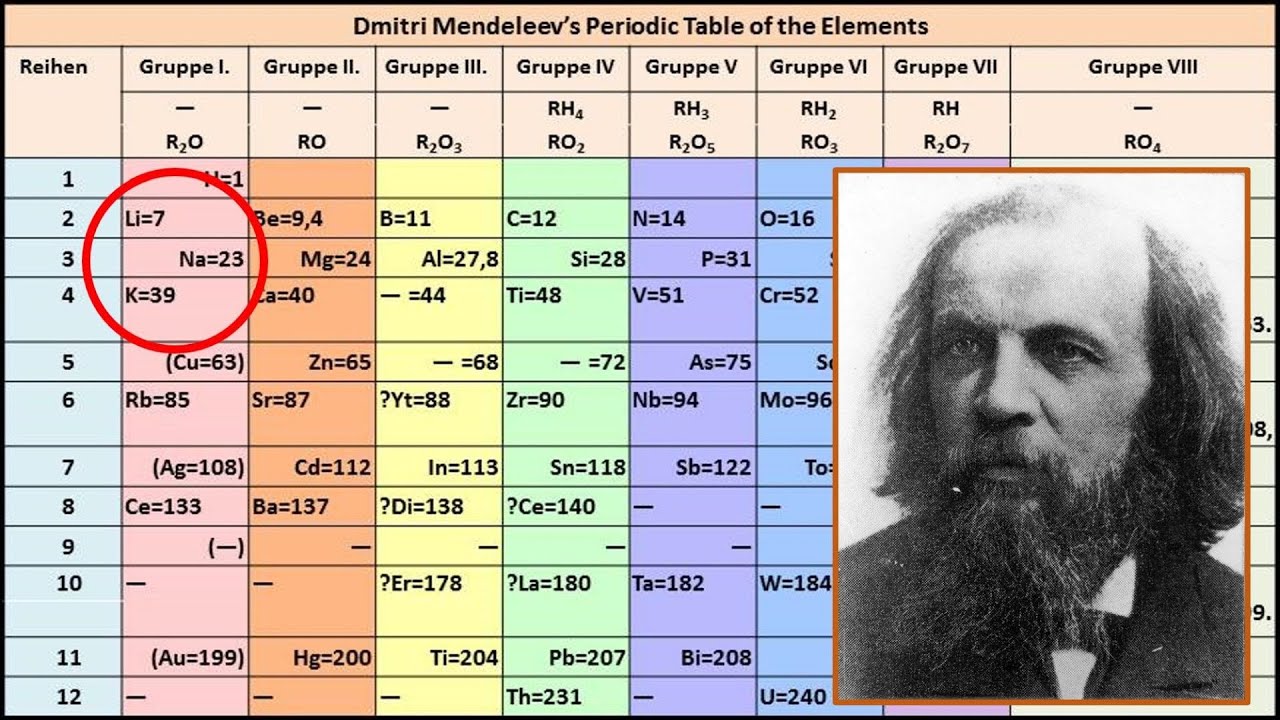
So convinced was he of the soundness of his periodic law that he left gaps for these elements in his table. Mendeleev arranged the periodic table in different-different ways. Soon, Mendeleev was predicting the properties of three elements – gallium, scandium and germanium – that had not then been discovered. Mendeleevs Periodic Table: Mendeleev was a Russian scientist. Now each chemical element had its number and fixed position in the table, and from this it became possible to predict its behaviour: how it would react with other elements, what kind of compounds it would form, and what sort of physical properties it would have. The discovery of Germanium later on, during his lifetime, proved him correct. Download royalty-free stock photos, vectors, HD footage and more on Adobe Stock. Eg: Mendeleev gave the names Eka Aluminium and Eka Silicon to those elements which were to be placed below aluminum and silicon respectively in the periodic table and predicted their properties. Mendeleev’s ideas, which built on the earlier work of French chemist Antoine Lavoisier in the previous century, totally changed the way chemists viewed their discipline. Search from thousands of royalty-free Mendeleev Periodic Table stock images and video for your next project.  
When he did this he noted that the chemical properties. It was perhaps the greatest breakthrough in the history of chemistry. Mendeleev arranged the elements in order of increasing relative atomic mass. He wrote down the sequence in such a way that they ended up grouped on the page according to known regularities or ‘periodicities’ of behaviour. He explicitly stated the periodic law: The properties of the elements are a periodic function of their atomic weights. On 17 February 1869, Russian chemist Dmitri Mendeleev jotted down the symbols for the chemical elements, putting them in order according to their atomic weights and inventing the periodic table. Dmitri Mendeleev, a Russian chemist, is widely known for the development of the periodic table and is regarded by the father of the modern periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
